Edition 15 | March 2026
Feature Article
Transforming Classrooms into Student-Owned Spaces
The Valency Trap!
Consider a Grade 7 class learning valency. Students are assessed at the end of the chapter. Most score well because they memorise:
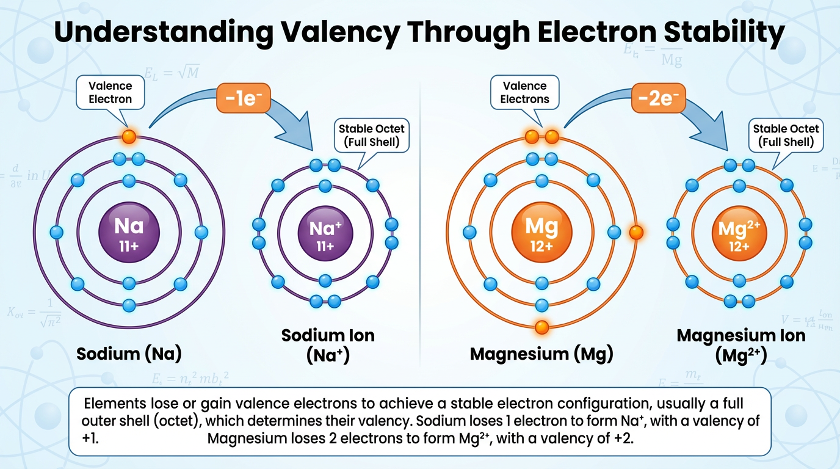
Sodium has valency 1, and Oxygen has valency 2
The test checks recall and procedural application. But what was the concept really testing?
It was meant to assess understanding of:
| 1 | Electron gain and loss |
| 2 | Stability of atoms |
| 3 | Formation of ions |
| 4 | Relationship between atomic structure and bonding |
When the Gap Appears
Now those same students move to Grade 8.
They are asked to:
| 1 | Write chemical formulae independently |
| 2 | Work with polyatomic ions |
| 3 | Understand ionic and covalent bonding |
Valency became a shortcut, not a story about electrons seeking stability.
Understanding Valency Through Electron Stability
What Happens If This Concept Does Not Develop?
If valency is not deeply understood:
Consequences of Weak Conceptual Foundation |
|---|
| Ionic bonding becomes confusing |
| Chemical equations are imbalanced |
| Periodic trends seem disconnected |
This is what we call failure in vertical learning, when a weak foundation blocks future understanding.
The issue is not effort. It is ownership.
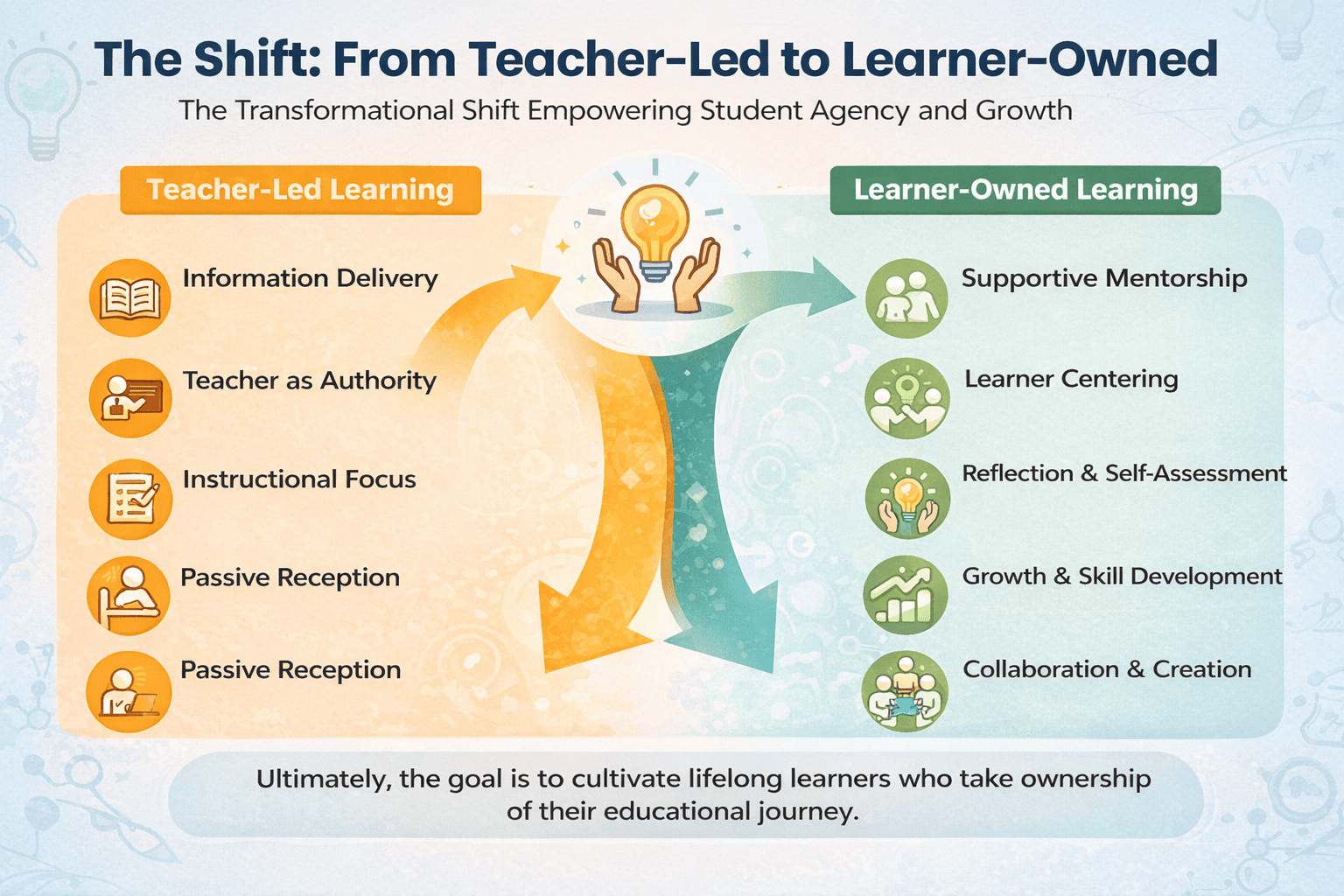
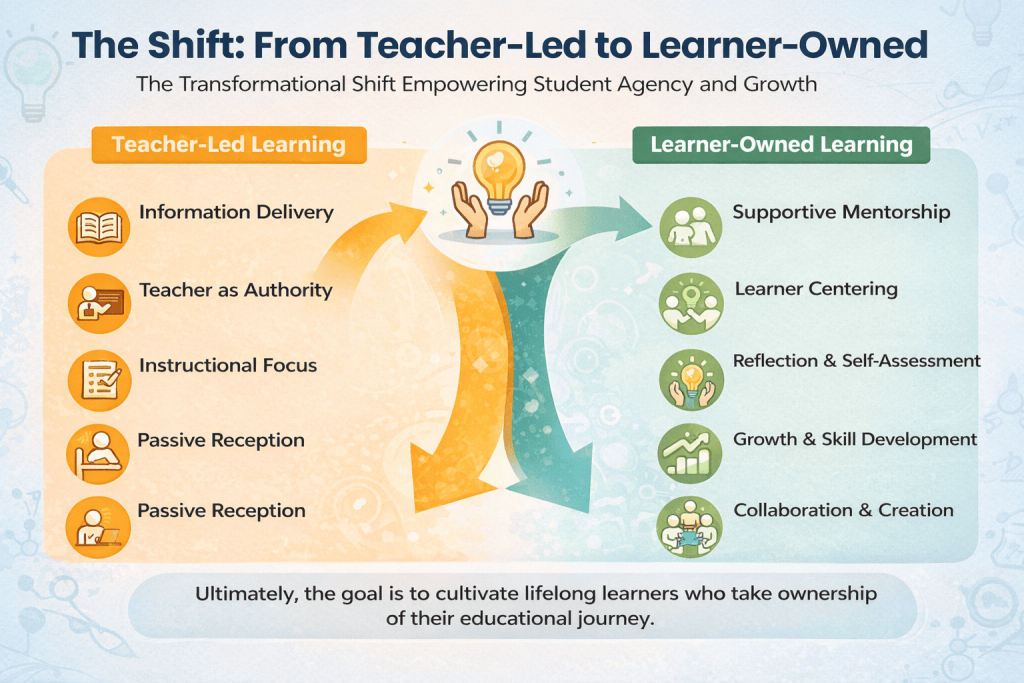
Shifting the Onus: From Teacher-Led to Learner-Owned
In many conversations about classroom reform, the spotlight remains on teaching strategies. But real change begins when students start examining their own understanding.
The Shift: From Teacher-Led to Learner-Owned
Consider the earlier classroom moment when students explored atomic number and electronic configuration before learning valence. When sodium was written as 2,8,1 and magnesium as 2,8,2, students were not immediately told their valencies. Instead, they were asked:
Guiding Questions for Student Reflection:
Questions That can Shift Thinking | |
|---|---|
| 1 | What pattern do you notice in the outermost shell? |
| 2 | Which atoms appear stable already? |
| 3 | What might sodium need to do to become stable? |
Some students explained stability through electron loss. Others simply linked valency to group number. That reflection pause revealed who truly understood and who was relying on surface patterns. At that moment, the responsibility subtly shifted. Instead of the teacher correcting and moving ahead, students analysed their own thinking. When later asked why magnesium forms MgCl₂ instead of MgCl, they reasoned through electron stability rather than applying a crossing rule.
This is the shift.
We aim to build independent individuals, yet often the full weight of learning rests on the teacher. When structured reflection is embedded at key conceptual stages, students begin asking themselves:
Student Self-Questioning:
Few Critical Self-Reflection Questions | |
|---|---|
| 1 | What do I understand about electron stability? |
| 2 | Where is my confusion? |
| 3 | What must I clarify before moving ahead? |
And ownership is what transforms classrooms from grade-focused spaces into growth-driven environments.
The Transformation in Learning Perspective
Reflection moves learning from:
What did the teacher teach?
to
What did I understand? What do I not yet understand? What will I do about it?
Here, the teacher becomes a facilitator. The learner becomes accountable.
This shift is demanding. It requires a lot of planning, patience, and reflection on the part of the teachers. It also demands that teachers take a backstep and allow productive struggle, withholding immediate correction and just being there for the students, leading them in the correct direction.
But it prepares students for life, not just exams.
Reflection Should Not Be an End Activity
If reflection happens only after the unit test, it is too late. Reflection must be embedded within the learning journey. This is the most crucial step. In the example stated above at the beginning of the text, if the gaps had been identified at a very early stage and closed by the learners, there would have been absolute clarity of the concept.
How Can This Be Practised in Class?
1. Mid-Lesson Concept Check Reflections
After teaching a key idea (e.g., valency and electron transfer), ask students:
Few Student Reflection Prompts | |
|---|---|
| 1 | What is one thing I am confident about? |
| 2 | What still confuses me? |
| 3 | What example can I create on my own? |
2. Misconception Analysis Routine
Present a common incorrect answer (e.g., MgCl instead of MgCl₂).
Ask:
Few Analytical Questions | |
|---|---|
| 1 | Why might someone think this is correct? |
| 2 | What assumption are they making? |
| 3 | What concept is missing? |
3. Learning Journals Linked to Vertical Growth
At the end of a lesson, students write:
Few Journal Prompts for Vertical Learning | |
|---|---|
| 1 | How will today’s learning help me next year? This process is also linked to the vertical mapping of the curriculum and identifying the power standards, which serve as an important aspect in building the concept. |
| 2 | Where might I use this concept again? |
4. Error-to-Action Reflection
After any assessment:
Reflection-to-Action Framework | |
|---|---|
| 1 | What mistake did I make? |
| 2 | Why did I make it? |
| 3 | What will I practise differently? |
The Real Reform
When students learn to:
| 1 | Identify their misconceptions |
| 2 | Take responsibility to close gaps |
| 3 | Connect present learning to future application |
Reforming education is not only about changing pedagogy. It is about cultivating learners who can think about their thinking and act on it. That is when independence truly begins.
The article is contributed by Ms. Anshika Saxena.
Instructional Coach, Science
Heritage International Xperiential School, Gurugram
Ms Anshika Saxena began her teaching journey in 2008. An engineer by profession and an educator by passion, she soon realised that her true calling was not in technical design, but in shaping young minds.For her, teaching has never been about the subject, it has always been about the teacher. When a teacher builds a genuine connection with students, learning naturally follows. Once trust is established, any concept can be taught, explored, and mastered.
She discovered that her Ikigai lies in the smiles of my students and in the lasting impact I create in their lives. Her focus extends beyond academic achievement to nurturing life skills like resilience, responsibility, critical thinking, reflection, and empathy because these are the tools that truly prepare students for the world beyond school.
She is a firm believer in continuous growth for educators as much as for students and see education as a powerful journey of connection, character building, and lifelong learning.
Are these principles already part of your teaching toolkit?
We’d love to hear your story!
Enjoyed the read? Spread the word
Interested in being featured in our newsletter?
Feature Articles
Join Our Newsletter
Your monthly dose of education insights and innovations delivered to your inbox!
powered by Advanced iFrame